How much should COVID-19 tests cost? DOH surveys market
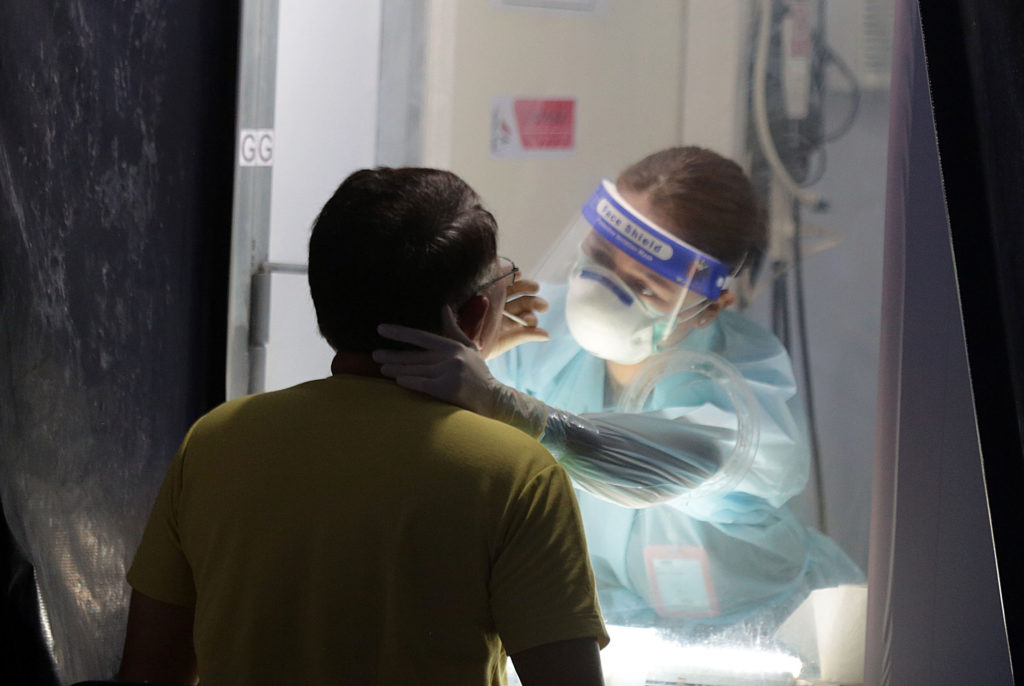
RICH MAN’S TEST The real-time polymerase chain reaction test for the new coronavirus costs P1,500 to P19,000 at public and private laboratories. The Department of Health is doing a market survey to bring the price of the test within the reach of poor COVID-19 patients. —GRIG C. MONTEGRANDE
The Department of Health (DOH) is doing market surveys to set affordable prices for coronavirus tests, including the real-time polymerase chain reaction (RT-PCR) test that it considers the “gold standard” in determining infection with the COVID-19 agent.
“We have already instructed our concerned offices to do the preparations like survey the price range of different testing methods currently on the market,” Health Undersecretary Maria Rosario Vergeire told an online briefing on Thursday, October 1, 2020.
Vergeire said the study would enable the DOH to issue specific guidelines in case the Office of the President approves the health agency’s recommendation for caps on the prices of coronavirus tests.
Price ceilings
Earlier, the DOH submitted a proposal to Malacañang for the issuance of an executive order that would set ceilings for the prices of coronavirus tests, after the agency had observed that the prices of tests in laboratories and hospitals varied.
The proposal won support from the medical community, which said current prices were beyond the reach of the poor. Doctors said it was unfair that people who could pay for tests got the results within hours, while the poor had to wait for weeks before they could get the results of their tests.
The DOH considers the PCR test the best for determining infection with SARS-CoV-2, the new coronavirus that causes the severe respiratory ailment COVID-19. But its price in public and private laboratories ranges from P1,500 to P19,000.
In its revised benefit package, Philippine Health Insurance Corp. pays only up to P3,409 if all the services for the testing were done by the laboratory or hospital.
Vergeire said the DOH had yet to complete the guidelines for the use of different tests for the COVID-19 virus.
“It has been stalled,” she said. The guidelines were supposed to have been issued on Sept. 11 but that the agency was still studying how the controversial antigen test could be used.
Earlier, the World Health Organization said the antigen test could not be used in specific circumstances, causing “a sudden change in the recommendation” of DOH experts, Vergeire said.
She said an antigen trial should be completed first before the guidelines could be released. The trial is being carried out in Baguio City.
Saliva test
Vergeire said experts were also evaluating other tests, including the saliva test.
The Philippine Red Cross and the Research Institute for Tropical Medicine are looking into the use of saliva test to detect the coronavirus, she said.
There are 105 PCR testing laboratories and 33 Genexpert labs in the country that have tested 3.5 million people, the DOH said.
314,079 cases
On Thursday, the DOH reported 2,415 additional coronavirus infections, raising the overall number of COVID-19 cases in the country to 314,079.
Of the new cases identified by 138 accredited laboratories, 930 were from Metro Manila, 238 from Cavite, 128 from Rizal, 123 from Laguna, and 103 from Negros Occidental.
The DOH said 771 more patients had recovered, bringing the total number of COVID-19 survivors in the country to 254,223. It said, however, that 59 more patients had died, raising the death toll to 5,562.
That left the country with 54,294 active cases, of which 86.6 percent were mild, 9.0 percent asymptomatic, 1.4 percent severe, and 3.1 percent critical.
Disclaimer: The comments uploaded on this site do not necessarily represent or reflect the views of management and owner of Cebudailynews. We reserve the right to exclude comments that we deem to be inconsistent with our editorial standards.
